Caution: None of the compounds here are intended for human consumption. These are compounds for researchers and experimental investigations by the scientific community.
Introduction
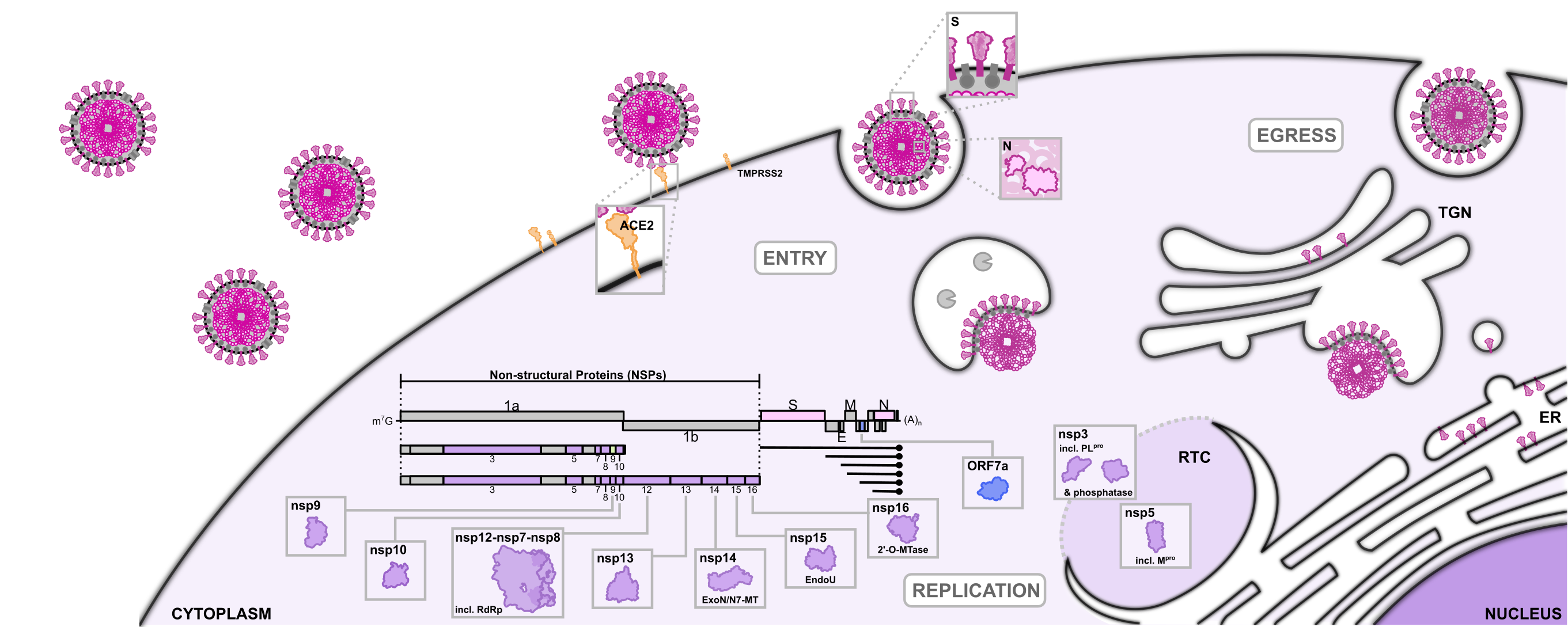
We have carried out a large number of ultra-large virtual screens against 17 important SARS-CoV-2 proteins, covering a total of 40 different sites among those 17 proteins.
The drug discovery platform which we have used is VirtualFlow.
We are making available the following datasets:
- An interactive table (for ultra-large virtual screen) containing the top 1000 hits. This interactive table allows to
- interactively filter the hits
- see important properties of each hit
- follow the link to the original catalog/or vendor of the compound
- visually explore the virtual hits in a 3D molecular viewer (Mol*) docked to the receptor structure
- The top 1 million hits (i.e. the top ~0.1 % of the virtual hits) downloadable as molecular database files (in the free DataWarrior format)
- Special datasets, such as lists of world-approved drugs among the virtual hits
To cite our work, please reference
A commercial service for ultra-large virtual screenings based on VirtualFlow is available via the company Virtual Discovery, Inc.